 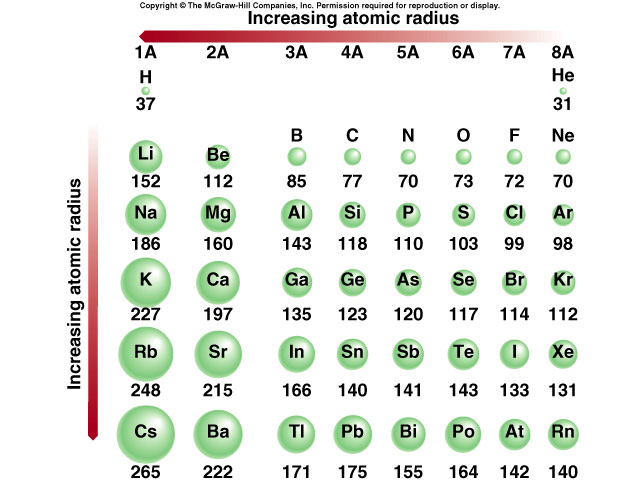 
The bond dipole moment uses the idea of electric dipole moment to measure the polarity of a chemical bond within a molecule. The percentage of ionic character in a compound can be estimated from dipole moments. Percentage of ionic character and charge distributionīased on Fajan's rules, it is expected that every ionic compound will have at least some amount of covalent character. It is found that the greater the possibility of polarization, the lower is the melting point and heat of sublimation and the greater is the solubility in non-polar solvents. Thus zinc (II) chloride ( Zn(II) 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 and Cl - 1s 2 2s 2 2p 6 3s 2 3p 6 ) is more covalent than magnesium chloride ( Mg(II) 1s 2 2s 2 2p 6) despite the Zn 2 + ion (74 pm) and Mg 2 + ion (72 pm) having similar sizes and charges.įrom an MO perspective, the orbital overlap disperses the charge on each ion and so weakens the electrovalent forces throughout the solid, this can be used to explain the trend seen for the melting points of lithium halides. < < and < < Įlectronic configuration of the cation: for two cations of the same size and charge, the one with a pseudo noble-gas configuration (with 18 electrons in the outer-most shell) will be more polarizing than that with a noble gas configuration (with 8 electrons in the outermost shell). The cation charge increases (size decreases) and on the right, the anion size increases, both variations leading to an increase in the covalency. The greater the positive charge, the smaller the cation becomes and the ionic potential is a measure of the charge to radius ratio. Large cations are to be found on the bottom left of the periodic table and small anions on the top right. Large charges: as the charge on an ion increases, the electrostatic attractions of the cation for the outer electrons of the anion increases, resulting in the degree of covalent bond formation increasing.This explains why for the common halides, iodides, are the most covalent in nature (I - 206 pm). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
